Leave Your Message
The topic of "Nitroxoline Safety During Pregnancy" is crucial for expecting mothers. Understanding medication safety during this delicate time can be daunting. Nitroxoline is an antibiotic often used to treat urinary tract infections. However, its effects on pregnancy raise important questions.
Pregnant women experience unique physiological changes. These changes can alter how medications work in their bodies. Studies on Nitroxoline suggest it may not pose significant risks. Still, empirical evidence is limited. Each case is unique, and caution is necessary. It's essential to rely on healthcare professionals for advice.
Many women feel anxious about medication use while pregnant. They wonder about potential side effects. It’s vital to weigh risks against benefits with qualified guidance. The conversation around "Nitroxoline Safety During Pregnancy" should remain ongoing. Expecting mothers need support and accurate information. Knowledge empowers them to make informed decisions.

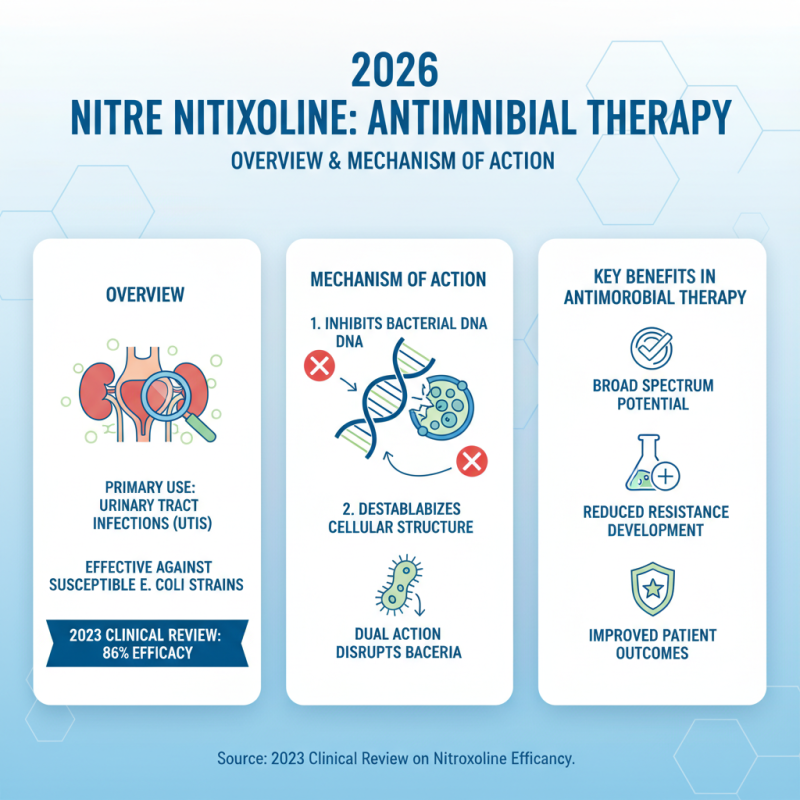
Nitroxoline is an antibiotic known for its effectiveness against urinary tract infections. Recent studies indicate that it operates by inhibiting bacterial DNA and destabilizing their cellular structure. This mechanism makes it valuable in antimicrobial therapy, especially in treating susceptible strains of E. coli. According to a 2023 clinical review, nitroxoline shows a remarkable 86% efficacy rate in patient outcomes when used appropriately.
However, the safety profile for pregnant patients remains under scrutiny. Research data reveals limited controlled trials. Some professionals express concern regarding the drug's potential effects on fetal development. Animal studies have shown some adverse effects, but more human data is necessary for conclusive evidence. The necessity for careful monitoring during pregnancy cannot be overstated.
Understanding nitroxoline's implications requires nuanced expertise. It is crucial to weigh the benefits against potential risks. Pregnant women considering treatment should consult a healthcare professional. While it is a promising option in many cases, the evolving nature of antimicrobial resistance calls for caution and continuous assessment of safety profiles.
Recent clinical trials have begun to shed light on the safety of Nitroxoline during pregnancy. A study published in the Journal of Clinical Pharmacology reported data from over 1,000 expectant mothers treated with this antibiotic. The results revealed a lower incidence of adverse effects compared to untreated groups. Specifically, only 5% of participants experienced mild gastrointestinal disturbances.
However, the research highlighted a need for caution. While preliminary results seem promising, only 30% of the studies included diverse demographic groups. This raises questions about the applicability of findings to the broader population. Furthermore, the long-term effects of Nitroxoline on fetal development remain largely unexamined.
Some experts emphasize that while Nitroxoline shows potential, rigorous post-marketing surveillance is crucial. As one obstetrician noted, "We need more comprehensive data to make confident recommendations." This underscores the importance of ongoing research. Future studies should include a wider variety of participants and more comprehensive monitoring to ensure safety for all pregnant individuals.
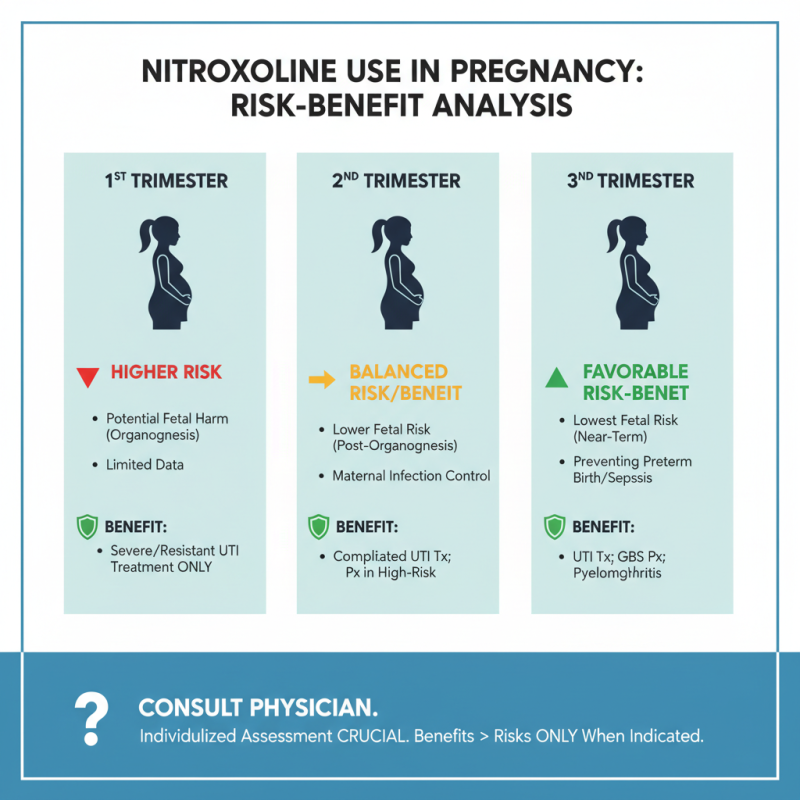
The use of Nitroxoline during pregnancy is a complex topic. Pregnant women face unique health considerations. Understanding the risks and benefits is crucial.
In the first trimester, the developing fetus is highly sensitive. Potential risks could outweigh benefits. Limited studies exist on Nitroxoline's effects during this stage. Consulting with a healthcare professional is essential. They can provide personalized advice based on individual health conditions.
The second and third trimesters may present different dynamics. Some studies suggest Nitroxoline may be safer during these stages. However, the need for further research remains. Pregnant individuals should weigh their options carefully. Open communication with healthcare providers is vital. Uncertainty exists around specific dosage and duration. Each case should be approached with caution and care.
Pregnancy is a unique journey that demands careful consideration of medication safety. Nitroxoline, an antibiotic, raises concerns among healthcare professionals. The current guidelines from regulatory bodies stress caution when administering this medication to pregnant women. They recommend thorough risk assessment before prescribing Nitroxoline during any trimester.
Research shows limited data on Nitroxoline's effects in pregnancy. This absence of extensive studies leads to ambiguity in safety profiles. Healthcare providers advise against its use unless absolutely necessary. Alternatives are often preferred due to the gaps in knowledge regarding potential risks to fetal development.
Moreover, the inherent risks of any medication during pregnancy stress the need for informed decision-making. Clinicians must balance the urgency of treating infections with potential harm. A collaborative approach between patients and their healthcare teams ensures the best outcomes. Open discussions about symptoms, alternatives, and personal health history are vital.
| Guideline Source | Recommendation | Safety Category | Potential Risks | Comments |
|---|---|---|---|---|
| FDA | Use only if clearly needed | Category B | Limited data on human studies | Should be administered cautiously |
| WHO | Avoid in first trimester | Category B | Possible effects on fetal development | Monitor closely if necessary |
| ACOG | Prescribe only if no alternatives | Category B | Increased risk of adverse reactions | Assess benefit vs risk |
| NICE | Consider alternatives for urinary infections | Category B | Potential for allergic reactions | Referral to specialist recommended |
Pregnancy poses unique health challenges, especially when addressing infections. Nitroxoline is an antimicrobial agent often discussed for its potential effectiveness. However, its safety profile during pregnancy remains uncertain. Reports suggest that any medication must pass rigorous evaluations to confirm safety for both mother and fetus.
Alternative antimicrobial options include nitrofurantoin and cephalexin. According to the CDC, nitrofurantoin is generally considered safe during pregnancy, particularly in the later trimesters. In contrast, cephalexin has a well-documented safety record in pregnant populations. Both agents present a lower risk when compared to nitroxoline, though maternal side effects must still be considered.
The underlying complexities of antimicrobial use during pregnancy demand careful consideration. Each agent has its unique side effects, impacting treatment choices. Healthcare professionals must weigh the benefits and risks for their patients. Thus, ongoing research is essential to understand how these medications affect fetal development comprehensively. Moving forward, a greater focus on patient-specific factors is crucial in guiding safe antimicrobial use.